|
11/18/2023 0 Comments Cdc up to date vaccine 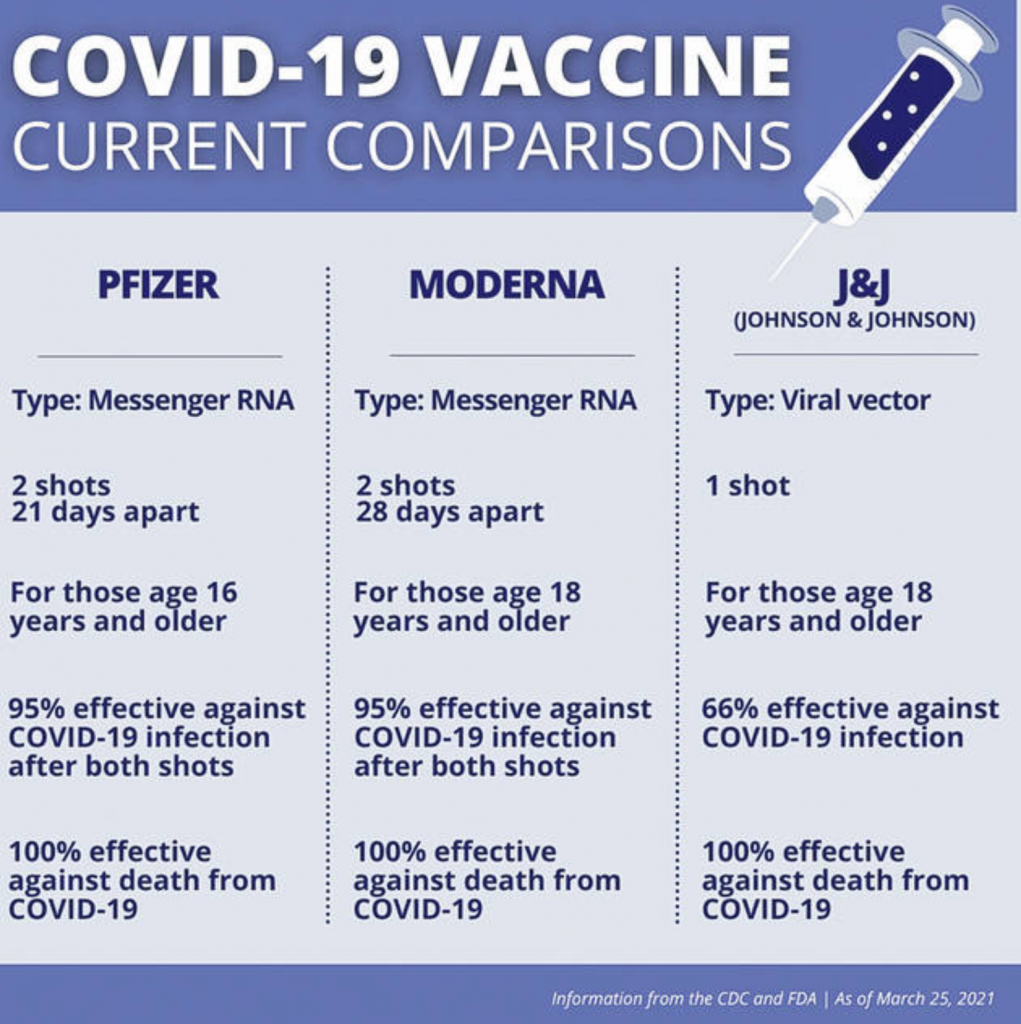
This page was reviewed on August 3, 2023. Health department websites and managers, and laws One-stop shop for official information about vaccines CDC: Centers for Disease Control and Prevention By The New York Times After three horrific years, in which Covid has killed more than one million. ACIP: Advisory Committee on Immunization Practices 30 20 10 2020 2021 2022 2023 Note: Data is through the week ending June 17, 2023. Recommended Immunization Schedule for Children and Adolescents Aged 18 Years or Younger Effectiveness of Up-to-Date COVID-19 Vaccination in Preventing SARS-CoV-2. Recommended Immunization Schedule for Adults Aged 19 Years or OlderĢ023 U.S. Monovalent Moderna and Pfizer-BioNTech COVID-19 vaccines are no longer authorized for use in US.įDA authorizes bivalent Pfizer-BioNTech COVID-19 vaccine as booster dose for certain children 6 months through 4 years of ageĢ023 U.S. 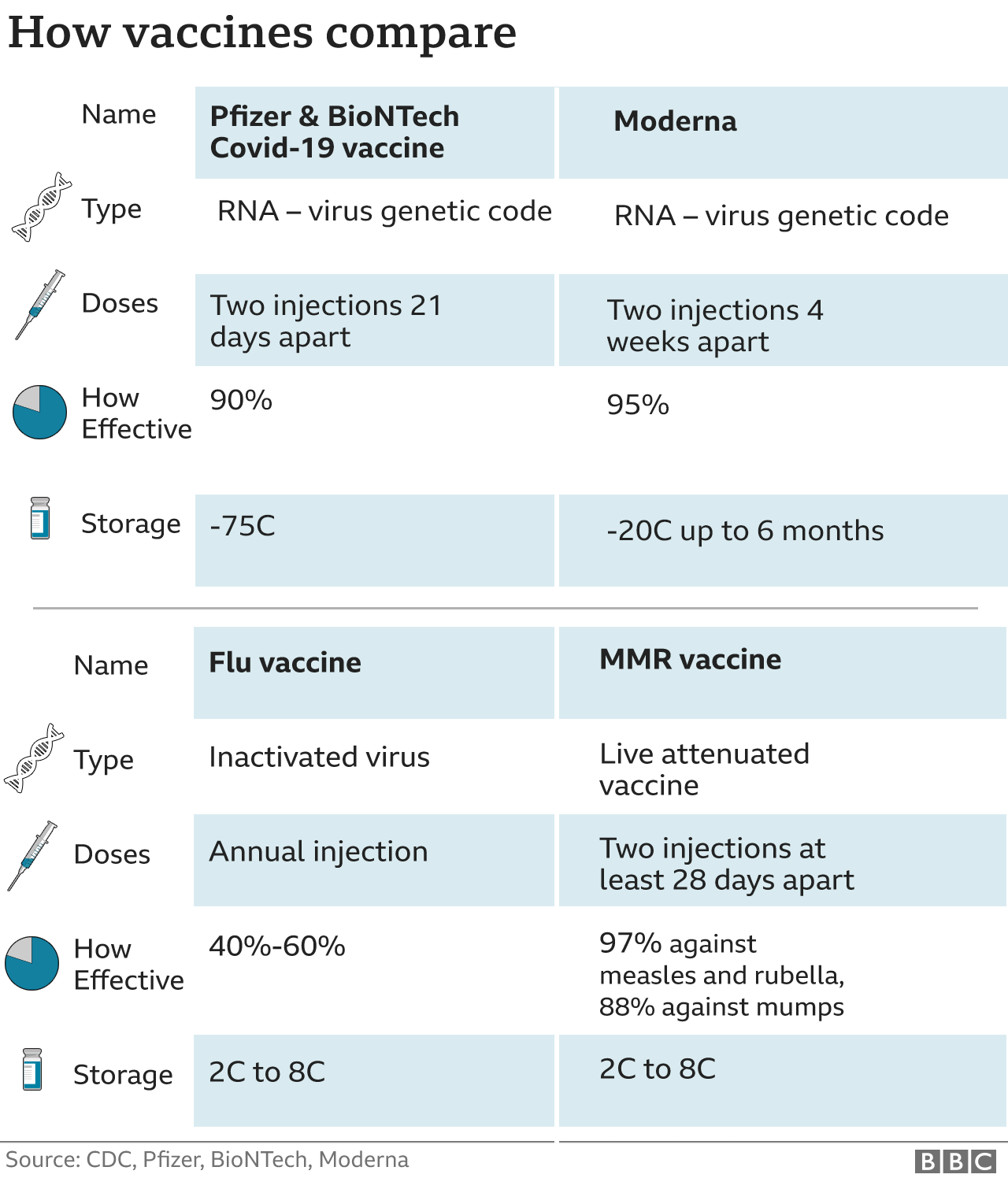
Bivalent vaccines are to be used for all doses given to individuals 6 months of age and older. Arexvy (GSK) is approved for individuals 60 years of age and older.įDA authorizes additional dose(s) of Pfizer-BioNTech COVID-19 Vaccine, Bivalent for individuals 6 months through 4 years of age with certain types of immunocompromise who have previously received three 0.2 mL doses (Pfizer-BioNTech COVID-19 Vaccine or Pfizer-BioNTech COVID-19 Vaccine, Bivalent)įDA approves Prevnar 20 (Pfizer) for infants and children, now approved for individuals 6 weeks and olderĬDC simplifies COVID-19 vaccine recommendations, allows older adults and immunocompromised adults to get second dose of the bivalent vaccine. Monovalent (original) COVID-19 mRNA COVID-19 vaccines no longer recommended for use in the United States.įDA amends EUAs of Moderna and Pfizer-BioNTech COVID-19 bivalent mRNA vaccines. Updated Pneumococcal Conjugate Vaccine Information Statement (VIS) postedįDA approves the first respiratory syncytial virus (RSV) vaccine. What You Need to Know Everyone aged 6 years and older should get 1 updated Pfizer-BioNTech or Moderna COVID-19 vaccine to be up to date. Updated Hepatitis B Vaccine Information Statement (VIS) posted Abrysvo is approved for individuals 60 years of age and older. 21, 2022 guidance with the change in language from "boosted" to "up to date" is available here.CDC adopts ACIP recommendation for the use of nirsevimab (Beyfortus, Sanofi), a long-acting monoclonal antibody product shown to reduce the risk of both hospitalizations and healthcare visits for RSV in infants by about 80 percentįDA approves Ebola vaccine (Ervebo, Merck) to include individuals 12 months of age and olderĬDC posts new Respiratory Syncytial Virus (RSV) Vaccine Information StatementĬDC posts updated Multiple Vaccines (“Your Child’s First Vaccines”) Vaccine Information StatementĪCIP recommendations on use of Respiratory Syncytial Virus (RSV) vaccines (Pfizer Abrysvo and GSK Arexvy) in older adultsįDA approves Cyfendus (Anthrax Vaccine Adsorbed, Adjuvanted - Emergent BioSolutions) a two-dose anthrax vaccine for post-exposure prophylaxis use for adults 18 through 65 yearsĬDC adopts 2023-24 ACIP flu vaccination recommendations, to be published later this summerĬDC recommends RSV vaccine for older adults using shared clinical decision-makingĪCIP interim recommendations for use of bivalent mRNA vaccines for persons aged 6 months – 5 years, adults 65 and older, and for those with immunocompromising conditionsįDA approves Abrysvo (Pfizer) respiratory syncytial virus (RSV) vaccine. An individual who has completed a primary series and is eligible for a booster, but has not received a booster, is not up to date.An individual who has completed a primary series and is not yet eligible for a booster is up to date. 
An individual who has completed a primary series and received a booster is up to date. 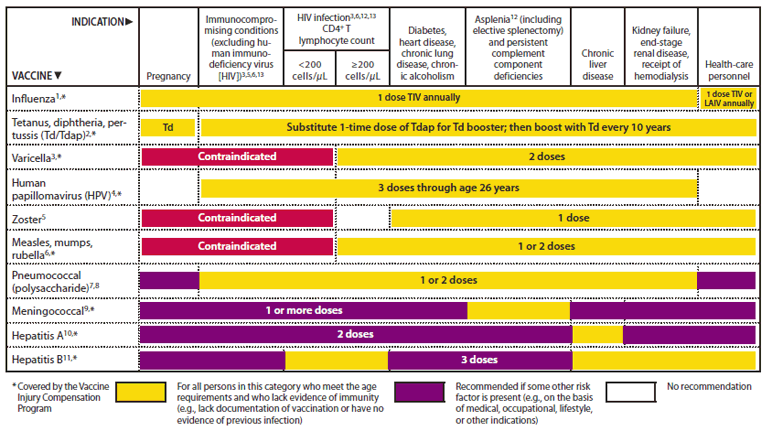
LeadingAge NY was able to clarify with the CDC this week that an individual is “up to date” if they have received all recommended doses of COVID-19 vaccine for which they are eligible. With the rise of the Omicron variant, the CDC began talking about vaccination status in a new way, using the phrases “up to date” and “not up to date.” This has created much confusion, particularly as guidance for isolation/quarantine and return to work has utilized these phrases. The new piece is the change in terminology from "boosted" to "up to date." The Centers for Disease Control and Prevention (CDC) has clarified the definitions of "up to date" and "not up to date" for the COVID-19 vaccine and booster mandates. CDC Clarifies Definitions of “Up to Date” and “Not Up to Date” for Vaccine Mandate 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
